Far More Transparency is Needed for COVID-19 Vaccine Trials
With vaccines against SARS-CoV-2, the virus that causes Covid-19, on the near-term horizon, U.S. policymakers are focusing on how to ensure that Americans get vaccinated. This challenge has been compounded by reports that White House officials are exerting undue influence over the agencies that would ordinarily lead such efforts, the Food and Drug Administration and the Centers for Disease Control and Prevention.
Modeling Contact Tracing Strategies for COVID-19 in the Context of Relaxed Physical Distancing Measures
Stanford Health Policy’s Joshua Salomon, a professor of medicine and senior fellow at the Freeman Spogli Institute for International Studies, and colleagues developed a mathematical model to examine the potential for contact tracing to reduce the spread of the coronavirus. They modeled contact tracing programs in the context of relaxed physical distancing under different assumptions for case detection, tracing coverage and the extent to which contact tracing can lead to effective quarantine and isolation.
Hepatitis C Treatment in Prisons — Incarcerated People’s Uncertain Right to Direct-Acting Antiviral Therapy
In a recent perspective published by the New England Journal of Medicine(NEJM), Stanford Law student Alexandra Daniels analyzed a growing body of federal litigation brought by prisoners with the hepatitis C virus (HCV) who are seeking access to treatment for their condition. With co-author and mentor, Law Professor David Studdert — also a professor of medicine at Stanford Health Policy — Daniels documented the dire public health problem of HCV in prisons.
Yifan Zhang
Encina Commons Room 222,
615 Crothers Way,
Stanford, CA 94305-6006
Yifan Zhang, PhD, is a Senior Research Scholar at Stanford University School of Medicine. Her research applies statistical and causal inference methods to large-scale population data to study health risks, health policy, and injury prevention. Her work focuses on how exposures—particularly firearm ownership and household dynamics—shape risks of mortality, including suicide and homicide. She has been a core member of the Study of Handgun Ownership and Transfer (LongSHOT) since its inception and has played a key role in its design and analysis.
Zhang collaborates with multidisciplinary teams across academia and government and leads the design and analysis of large longitudinal studies using complex administrative data. Her research also examines drivers' crash risks, physician behavior following malpractice claims, and health outcomes in vulnerable populations.
She received her PhD and MSc from Harvard University and her BSc in Actuarial Science from the University of Hong Kong.
Response to COVID-19 in Taiwan Big Data Analytics, New Technology, and Proactive Testing
Taiwan is 81 miles off the coast of mainland China and was expected to have the second highest number of cases of coronavirus disease 2019 (COVID-19) due to its proximity to and number of flights between China. The country has 23 million citizens of which 850 000 reside in and 404 000 work in China. In 2019, 2.71 million visitors from the mainland traveled to Taiwan. As such, Taiwan has been on constant alert and ready to act on epidemics arising from China ever since the severe acute respiratory syndrome (SARS) epidemic in 2003. Given the continual spread of COVID-19 around the world, understanding the action items that were implemented quickly in Taiwan and assessing the effectiveness of these actions in preventing a large-scale epidemic may be instructive for other countries.
New Book Highlights Policy Initiatives and Economic Research on Healthy Longevity Across Asia
Rapid population aging is transforming societies around the world, and the Asia-Pacific region is on the frontlines of this unprecedented demographic shift. Life expectancy in Japan, South Korea, and much of urban China has now outpaced that of the United States and other high-income countries. With this triumph of longevity, however, comes a host of health, social, and economic challenges.
Longer lifespans will necessitate working to older ages, “but extending work lives will only be feasible if the added years are healthy ones, and will only be equitable if the least advantaged also benefit from healthy aging,” writes APARC Deputy Director and Asia Health Policy Program Director Karen Eggleston in her new book, Healthy Aging in Asia. “The great blessing of longer lives dims when clouded by pain, disability, and loss of dignity.”
[Listen to our conversation with Eggelston about the book and continue reading below. To receive stories like this directly in your inbox sign up for APARC newsletters]
Shorenstein APARC · Healthy Aging In Asia | Karen Eggleston
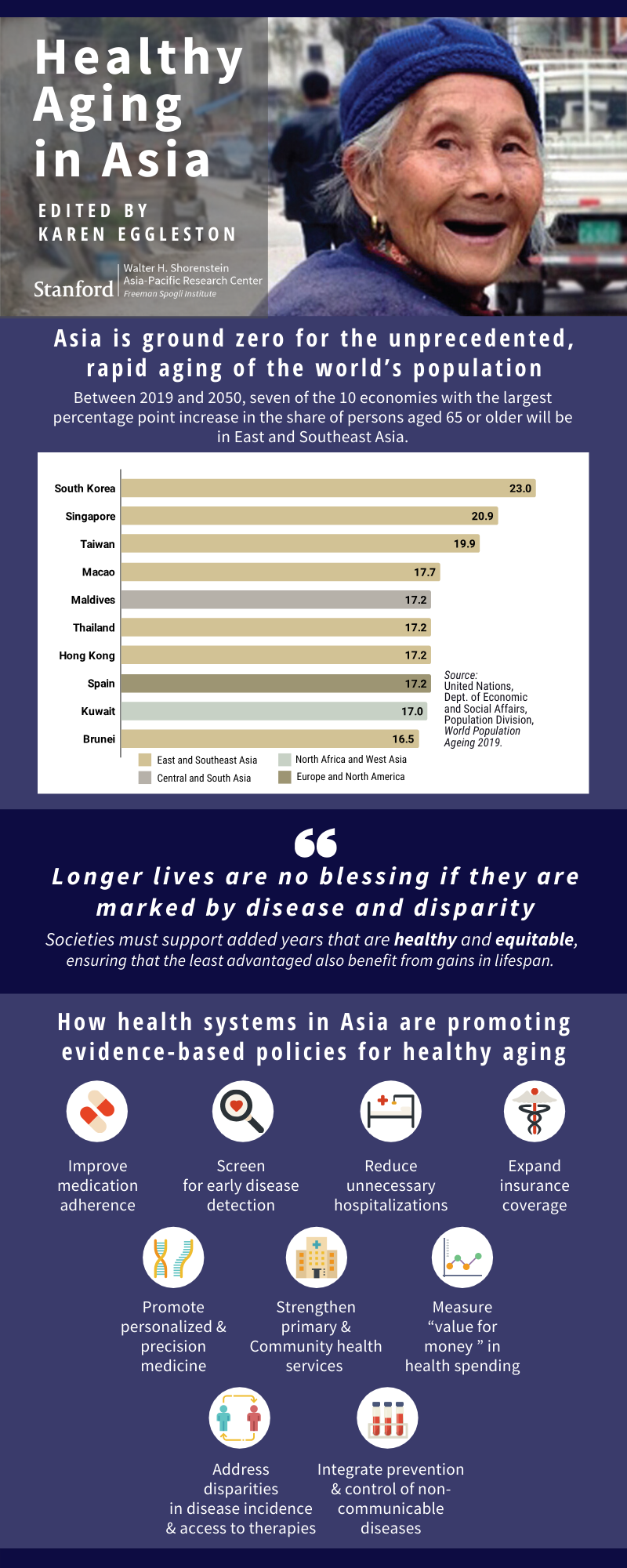
Societies around the world must reduce disparities in health outcomes and address the older age-associated rise in the burden of noncommunicable diseases (NCDs) such as diabetes, hypertension, and cancer. Indeed, the COVID-19 pandemic has underscored the vulnerability of older adults suffering from NCDs to newly emerging pathogens and the importance of building long-term, resilient health systems.
How are health systems in Asia promoting evidence-based policies for healthy aging? What strategies have they used to prevent NCDs, screen for early disease detection, raise the quality of care, improve medication adherence, reduce unnecessary hospitalizations, and increase “value for money” in health spending?
The concise chapters in Healthy Aging in Asia examine these questions, covering multiple aspects of policy initiatives and economic research on healthy longevity in diverse Asian economies — from cities such as Singapore and Hong Kong to powerhouses such as Japan, India, and China — as they transform their health systems to support wellbeing in older age. Eggleston edited and contributed multiple chapters to this new volume, now available via Brookings Institution Press. This publication is part of APARC’s in-house series with the Brookings Institution.

Karen Eggleston
Read More
Asia health policy expert Karen Eggleston’s new volume, ‘Healthy Aging in Asia,’ examines how diverse Asian economies – from Singapore and Hong Kong to Japan, India, and China – are preparing for older population age structures and transforming health systems to support patients who will live with chronic disease for decades.
Is the Coronavirus as Deadly as They Say?
Stanford Health Policy's Eran Bendavid and Jay Bhattacharya write in this Wall Street Journal editorial that current estimates about the COVID-19 fatality rate may be too high by orders of magnitude.
"If it’s true that the novel coronavirus would kill millions without shelter-in-place orders and quarantines, then the extraordinary measures being carried out in cities and states around the country are surely justified. But there’s little evidence to confirm that premise—and projections of the death toll could plausibly be orders of magnitude too high.
"Fear of Covid-19 is based on its high estimated case fatality rate — 2% to 4% of people with confirmed Covid-19 have died, according to the World Health Organization and others. So if 100 million Americans ultimately get the disease, 2 million to 4 million could die. We believe that estimate is deeply flawed. The true fatality rate is the portion of those infected who die, not the deaths from identified positive cases."
"The latter rate is misleading because of selection bias in testing. The degree of bias is uncertainbecause available data are limited. But it could make the difference between an epidemic that kills 20,000 and one that kills 2 million. If the number of actual infections is much larger than the number of cases—orders of magnitude larger—then the true fatality rate is much lower as well. That’s not only plausible but likely based on what we know so far."

Michelle Mello Answers Questions About the Federal Rollout of the Coronavirus Test
Controversies over the lack of diagnostic testing for the COVID-19 virus have dominated U.S headlines for weeks. Technical challenges with the first test developed by the Centers for Disease Control and Prevention (CDC) left the nation with minimal diagnostic capacity during the first few weeks of the epidemic, according to a new paper published today in the Journal of the American Medical Association by Michelle Mello, a professor of medicine at Stanford Health Policy and professor of law at Stanford Law School.
On February 29, the Food and Drug Administration (FDA) began allowing high-complexity labs across the country to use tests they developed in-house. On March 5, the Stanford Clinical Virology Lab deployed its own test for patients at Stanford Health Care and Stanford Children’s Health.
We asked Mello to answer some questions about the federal rollout of diagnostic testing.
You write that in the early stages, COVID-19 “spread beyond the nation’s ability to detect it.” Is there anything the U.S. government could or should have done weeks ago to get out ahead of the spread?
Adopting broader testing criteria and allowing use of a wider range of tests would have been helpful in identifying the first U.S. cases and containing the spread. Manufacturing problems like the one that arose with CDC’s test are always a risk, but the fact that CDC put all its eggs in that one basket made the manufacturing snafu highly consequential.
Also, the public messaging from Washington about the seriousness of the problem has been neither consistent nor accurate, and I worry it may have led Americans to take fewer steps to prevent community transmission than we should have. Containment was not “pretty close to airtight.” A vaccine was never going to be ready in “three to four months,” as the Trump administration claimed. The case fatality rate is not “way under 1 percent.” Part of the problem here is that as the stock market continues to plunge, the president and the task force he appointed appear to be more concerned about calming investors than stopping the virus.
We seem to be between a rock and a hard place: You write that remedying gaps in testing is imperative, yet “more testing is not always better.” How do we determine the happy middle ground?
First, the testing criteria have to be calibrated to our actual testing capacity. You can’t announce that any American who wants a coronavirus test can get one and then, within hours, announce that there aren’t enough test kits to make that possible. High priorities for testing include patients with serious, unexplained respiratory illness and contacts of known cases. From there, testing can be expanded, beginning with other high-risk groups, as capacity permits.
Second, we should consider unintended side effects of mass testing. The problem with this virus is that it doesn’t have signature symptoms. It looks like the common cold or the flu. If everyone with a cough or fever, or who has been around someone with a cough or fever, shows up in their doctor’s office demanding a test, it will quickly overwhelm care facilities that should be focusing on patients with a higher likelihood of being infected or and those who are infected and are seriously ill. It may also work against the social distancing measures that public health officials are trying to encourage, because crowded waiting rooms may spread the virus.
The CDC announced Monday it now has the testing capacity in 78 state and local public health labs across 50 states to test for the virus. There are now 75,000 lab kits cumulatively to test for COVID-19 with more coming on board by mid-March. But is there anything we could have done to roll this out earlier?
The alternative would have been to allow laboratories to deploy their own tests from the beginning, using the primers and protocols made publicly available by the World Health Organization. That’s what other countries have done. RT-PCR is a mature technology and high-complexity labs around the country are well-qualified to conduct this type of testing.
There is a public health argument for not going that route: perhaps those labs wouldn’t have done as good a job as CDC’s own lab and the state labs that it handpicked early in the outbreak. What if there were erroneous test results? We could miss cases, or we could put people into isolation, with huge social consequences, based on false-positive results. There is also a worry that some labs aren’t consistent about reporting positive test results to CDC, and underreporting could compromise disease surveillance efforts.
The counterargument is that high-complexity labs have that certification for a reason—they’re good at what they do. And of course, surveillance is also compromised when you miss cases because you don’t test.
You write in your paper that testing for COVID-19 “highlights a controversial area of public policy—the regulation of laboratory-developed tests—in which there has long been tension between the goals of access and quality.” Who should be in charge of regulating these tests?
Laboratory-developed tests are largely unregulated outside of emergencies. The FDA proposed draft guidance in 2014 that, if implemented, would have required labs to make certain showings to FDA about tests they developed in-house, with the particular evidence calibrated to the risks involved in having a wrong test result. Contrary to President Trump’s claim that an Obama-era policy constrained coronavirus testing, the guidance did not relate to emergency situations. During declared emergencies, another statute and set of regulations apply, and the FDA has broad discretion to allow or disallow use of novel diagnostics and therapies as emergency countermeasures.
As a general matter, it makes good sense to require labs to submit evidence that their in-house tests work. It’s odd that laboratory-developed tests are carved out of requirements that apply to other kinds of medical devices. It’s also sensible that our legal framework allows FDA’s regular rules to be relaxed during emergencies so we can tailor our response to the difficult and changing circumstances.
You write that diagnostic testing is critical to an effective response to the novel coronavirus. What sort of policies and guidelines should be put into place to prevent such a sluggish rollout during an emerging epidemic the next time one comes around?
The legal framework for an effective emergency response is in place. Because giving agency heads the discretion to act as potentially unforeseeable circumstances require is a linchpin of this legal framework, it only works if leaders make smart choices. Every emergency is different, and there is a danger of Monday-morning quarterbacking. But we should learn from every misstep we make, and I think the lesson here is to make better use of already developed networks of highly qualified labs to make sure we have adequate testing capacity to isolate cases and trace their contacts very early in an outbreak.
What are some innovative approaches we could be taking to speed up testing for those who really need it?
The South Koreans have set up drive-through testing stations in parking lots to avoid concentrating crowds of people indoors. Of course, that requires that you have plenty of test kits, which we don’t yet – but we should also be thinking about creative ways to address the epidemic. For example, how could video calls be used to monitor the health of people confined at home after being exposed to the virus? How can social media be used to connect neighbors to help one another when some are isolated at home? Hopefully we can find new ways for technology to bring us together when pathogens drive us apart.

Task Force: Not Enough Evidence to Recommend Screening for Cognitive Impairment in Elderly
More evidence-based research is needed before the U.S. Preventive Services Task Force can recommend that clinicians screen their older patients for cognitive impairment such as dementia and Alzheimer’s disease.
Cognitive impairment is a growing public health dilemma that affects millions of Americans as they age. The Global Burden of Disease study shows that Alzheimer’s rose from the 12th most burdensome disease or injury in the United States in 1990 to the 6th in 2016.
Medical experts who were commissioned to conduct an evidence report for the Task Force projected that the burden of Alzheimer’s disease is expected to grow to 13.8 million U.S. residents by 2050 — or nearly 3.3% of the projected U.S. population by that year.
Their findings, the Task Force recommendation statement and several accompanying editorials were all published Tuesday in the Journal of the American Medical Association.
The symptoms of cognitive impairment can range from problems with memory and language, to learning new things or making decisions that affect everyday life.
“Early identification of cognitive impairment through screening would ideally allow patients and their families to receive care at an earlier stage in the disease process, potentially facilitating discussions regarding health, financial, and legal decision-making while the patient still retains decision-making capacity,” the authors of the Task Force evidence report wrote.
But after reviewing some 287 studies including more than 285,000 older adults, the Task Force determined there wasn’t sufficient evidence about the benefits or harms of screening adults 65 and older who do not have signs or symptoms. The Task Force also did not find adequate evidence that screening for cognitive impairment improves decision-making or planning by patients, caregivers or doctors.
At the same time, there is little evidence on potential harms of screening, such as depression, anxiety or lower quality of life.
“Given the burden of dementia and the intense public interest in preventing cognitive impairment, the lack of progress is disheartening,” Carol Brayne, MD, with the Department of Public Health and Primary Care at the University of Cambridge in the UK, wrote in an accompanying JAMA editorial to the Task Force evidence report.
But, she added, “Political considerations and pressure from commercial interests and patient advocacy groups notwithstanding, public policies for dementia screening should be supported by evidence.”
The Task Force — an independent panel of national experts in prevention and evidence-based medicine — encourages clinicians to remain alert for early signs of symptoms of cognitive impairment, while calling for more research on the detection of dementia.
“Research is especially needed on whether screening and early detection of cognitive impairment helps patients, caregivers, and doctors make decisions about health care or plan for the future,” said Douglas K. Owens, chair of the Task Force and the director of Stanford Health Policy. “We share the frustration of clinicians who want to offer something that could help patients prevent cognitive impairment. We hope that additional research will enable us to know whether that’s possible.”
The most commonly used screening tests include the Mini-Mental State Examination as well as the clock-drawing test. Screening tests involve asking patients to perform a series of tasks that asses one or more aspects of cognitive functions. The USPSTF concluded that more research is needed to know whether such screening tests can lead to interventions that help prevent or improve cognitive impairment.




